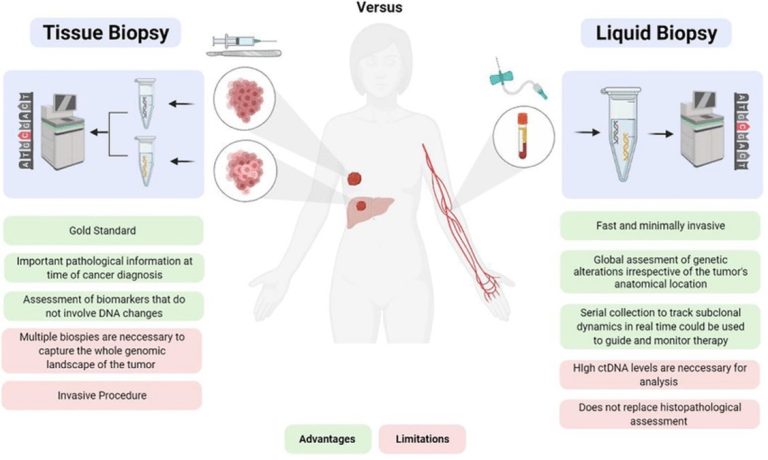
Limitations of tissue-based next-generation sequencing
Tissue-based next-generation sequencing (NGS) testing is the current standard of care in solid tumors to identify mutations that can be treated with approved biomarker-guided therapies, which lead to better clinical outcomes compared with non-targeted therapies. However, there are potential limitations in performing NGS testing using tumor tissue. First, it involves invasive sampling procedures and some patients may be medically unfit for such procedures or tumors may not be easily accessible, particularly in patients with metastatic cancer. In addition, there are also associated risks and costs to these biopsies. Second, NGS tests typically require a certain amount of material and quality, which may not always be adequate in biopsies. It is well known that a fraction of tissue-based NGS tests are unable to proceed due to insufficient or low-quality material. Third, tissue-based sequencing may not fully capture the intra- and inter-tumor heterogeneity, which may impair accurate treatment selection.
Advantages of blood-based circulating tumor DNA testing
Research has shown that tumors can release small fragments of DNA known as circulating tumor DNA (ctDNA) into the bloodstream. Liquid biopsy NGS, which utilizes samples from bodily fluids like blood samples, enables comprehensive profiling of ctDNA to identify tumor-derived actionable mutations as well as genomic signatures such as tumor mutational burden (TMB) and microsatellite instability (MSI) to guide therapy selection in solid tumors, without the need for invasive tissue biopsies. Hence, blood-based ctDNA sequencing is becoming more widely used for genomic profiling of cancers, offering several advantages over tissue-based NGS. In addition to being non-invasive, this approach provides easier access, ability to conduct serial testing and quicker turnaround times. It also has a lower failure rate, better detection of resistance mutations, and offers a more comprehensive representation of tumor heterogeneity.
Circulating tumor DNA can be used to guide therapy selection in clinical settings
Multiple prospective and retrospective studies have reported high positive predictive values between blood ctDNA and tissue-based analyses in advanced cancers. The growing body of evidence demonstrating the clinical utility of ctDNA in guiding therapy selection has led European Society for Medical Oncology (ESMO) to recommend that ctDNA tests may be routinely used when faster results will be clinically important, or when tissue biopsies are not possible or inappropriate. However, ESMO also recommends that reflex tumor testing should be considered following a non-informative ctDNA result, due to possible false negative results associated with ctDNA tests (when low ctDNA levels or fraction prevent detection of the mutations).
Blood-based ctDNA can be used for longitudinal monitoring in advanced cancers
Due to the short half-life of ctDNA (less than 2 hours in cancer patients) and the ease of repeated blood sampling, blood-based ctDNA can be used for real-time monitoring of disease during therapy in advanced cancers. Multiple studies have shown that ctDNA levels correlate with treatment response across multiple cancer types and may identify responses earlier than clinical or radiological detection, with ctDNA levels dropping within weeks of starting therapy in patients responding to chemotherapy, targeted therapy or immunotherapy. Sequential blood-based ctDNA analysis can also be utilized to monitor emergence of resistance mutations, such as ESR1 mutations that can develop during aromatase inhibitor treatment, to guide therapy adjustments. Advanced breast cancer patients who switched to alternative therapies upon early detection of ESR1 mutations like elacestrant (approved by the US FDA in 2023 for pre-treated ER-positive, HER2-negative, ESR1-mutated advanced or metastatic breast cancer) benefit from improved median progression-free survival. Notably, ESMO recommends that ESR1 mutations should preferentially be tested using ctDNA in breast cancer patients, with tissue biopsies often reporting false negative results. Although tissue-based testing remains the preferred approach, these recommendations highlight the growing evidence supporting the clinical utility of ctDNA to guide or adjust therapy selection in advanced cancers. It can serve as an alternative or complement to tissue-based testing, especially when tissue biopsies are inadequate or time is crucial.
Canary Oncoceutics has a steadfast commitment to three fundamental pillars: advancing scientific knowledge, fostering collaboration, and ultimately, enhancing the lives of cancer patients worldwide. From cutting-edge research to impactful clinical advancements, Canary Oncoceutics aims to illuminate the transformative potential of tailored cancer treatments. Join us on this journey towards a future where every cancer patient receives personalized, effective treatment tailored to their unique needs.