Actionable mutations in cancers
Cancer was once considered a single disease. However, differences in treatment responses and diverse effects among the same cancer types have highlighted the underlying differences in cancers. In light of this, cancers are now categorised into different subtypes based on histology and driver mutations, with clinical management varying depending on the specific subtype. There are four major classes of mutations: single nucleotide variants (SNVs), copy number alterations (CNAs), rearrangements, and insertions and deletions (indels). A single tumor can have multiple types of mutations in multiple genes. Importantly, certain mutations are actionable, i.e. can be targeted with specific therapies (targeted therapies and immunotherapies) to improve patient outcomes. Hence, various molecular testing strategies have been developed to detect biomarkers associated with these therapies.
Detecting mutations with single-gene and multiplex hotspot tests
Historically, a single-gene testing approach, such as using polymerase chain reaction (PCR) to test EGFR mutations or fluorescence in situ hybridization (FISH) to detect ALK rearrangements, has been used to detect mutations due to its lower cost, simpler workflow and ability to produce rapid results. Since cancer arises from multiple gene mutations, single-gene testing may not always yield findings in first instance. When this happens, additional testing may be required, leading to a delay in identifying the right treatment and increasing the testing costs. The situation is further compounded in cases with insufficient samples from initial biopsy (acquiring biopsies can be challenging, particularly in certain cancer types such as non-small cell lung cancer). Therefore, most laboratories have shifted from single-gene testing to multiplex hotspot testing. This approach, using either PCR, FISH or next-generation sequencing (NGS) techniques, targets several actionable mutations at hotspot regions located within multiple genes in a single assay. Although this approach is an improvement from single-gene testing, it cannot detect all four major classes of mutations in a single workflow. Furthermore, multiplex hotspot testing focuses on a predefined list of genes that are selected because of their known associations with certain cancer types. While this approach is effective for known mutations, it may miss out on emerging biomarkers associated with disease progression or response to treatment, as well as other important mutations in genes that are not included in the panel.
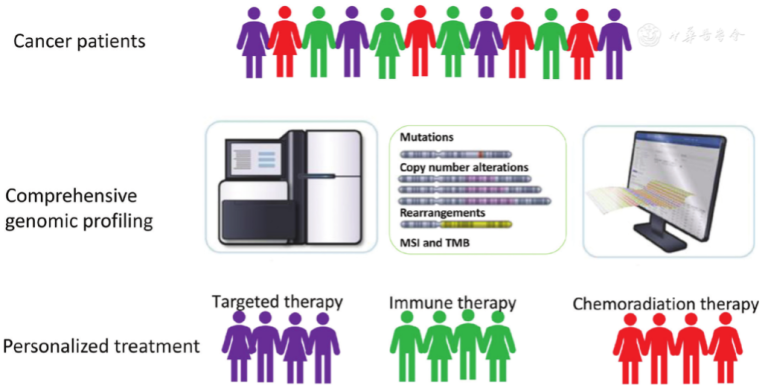
Comprehensive genomic profiling detects hundreds of biomarkers in a single assay
Due to the rapidly growing number of approved targeted therapies and immunotherapies in recent years, analysing a broader panel of genes for multiple classes of mutations has become increasingly important. Hence, NGS-based comprehensive genomic profiling (CGP) has emerged as an important approach to provide a holistic view of the genomic characteristics within a tumor. CGP is able to assess hundreds of prevalent and rare biomarkers, as well as genomic signatures such as tumor mutational burden (TMB), microsatellite instability (MSI), homologous recombination deficiency (HRD) and loss of heterozygosity (LOH) from a single sample. Multiple studies have demonstrated the ability of CGP to identify more actionable genomic alterations across different cancer types than single-gene or multiplex hotspot tests, eliminating the need for sequential testing. Aside from having high specificity and sensitivity, CGP also offers other advantages such as shorter time to actionable test results, conserves precious biopsy samples and reduces the need for re-biopsy. CGP is particularly beneficial for patients with unknown primary, rare, advanced or metastatic cancers, whereby single-gene and multiplex hotspot tests may not be able to fully capture the complexity of the disease. CGP can help to detect rare or novel mutations in these cancers, that could potentially open up new treatment pathways or identify innovative clinical trials that might be a good match for these patients based on their tumors’ genetic profile. Since the cost of NGS has dramatically decreased over the years, it is not surprising that more and more studies are now demonstrating the value of integrating CGP into new standard of care for cancer patients to improve patient outcomes.
Canary Oncoceutics has a steadfast commitment to three fundamental pillars: advancing scientific knowledge, fostering collaboration, and ultimately, enhancing the lives of cancer patients worldwide. From cutting-edge research to impactful clinical advancements, Canary Oncoceutics aims to illuminate the transformative potential of tailored cancer treatments. Join us on this journey towards a future where every cancer patient receives personalized, effective treatment tailored to their unique needs.