Important role of biomarkers in cancer management
According to National Cancer Institute, a biomarker is “a biological molecule found in blood, other body fluids, or tissues that is a sign of a normal or abnormal process, or of a condition or disease”. The use of biomarkers has revolutionized cancer management, with biomarkers now being used to help determine risk of developing cancer, diagnose types of cancer, identify suitable treatment, monitor treatment response and predict likelihood of disease progression or recurrence. Advancement in molecular diagnostics have led to characterization of molecular biomarkers with higher specificity and sensitivity than traditional antigen-based cancer biomarker such as carcinoembryonic antigen (CEA), alpha-fetoprotein (AFP) and prostate-specific antigen (PSA). Molecular biomarkers have thus become an integral component of precision medicine, enabling oncologists to tailor treatment based on the unique molecular profile of each patient’s tumor and promising better treatment outcomes.
Targeted therapies improve patient outcomes
Trastuzumab (Herceptin), which targets HER2 receptors, was approved by the U.S. FDA in 1998 to treat HER2-positive metastatic breast cancer, making it the first targeted therapy that requires biomarker testing to determine patients’ suitability for this therapy (by looking at HER2 expression). Prior to trastuzumab’s approval, patients with HER2-positive metastatic breast cancer were treated with conventional chemotherapy regimens and had higher rates of disease recurrence and mortality. A pivotal Phase III trial involving 469 HER2-positive metastatic breast cancer patients demonstrated that addition of trastuzumab to chemotherapy improved response rates (50% vs. 32%, P<0.001), extended time to progression (median, 7.4 vs. 4.6 months; P<0.001), lowered rate of death at 1 year (22% vs. 33%, P=0.008), led to longer survival (median survival, 25.1 vs. 20.3 months; P=0.01), and had a 20% reduction in the risk of death. Since then, more targeted therapies have been approved by the U.S. FDA for a broad range of solid tumors and hematologic malignancies.
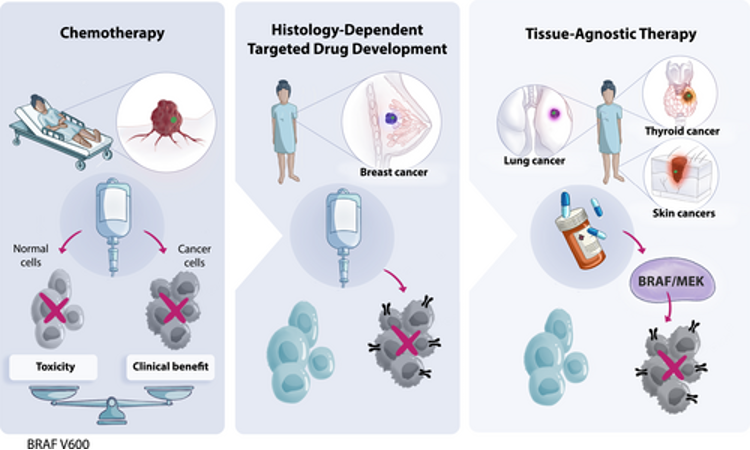
Tumor-agnostic therapies represent a paradigm shift in oncology
Historically, cancer therapies have always been approved for specific cancers based on their origin or location. This changed in 2017, when the US FDA approved pembrolizumab (Keytruda) based on a common molecular biomarker rather than the location in the body where the tumor originated, making it the first tumor-agnostic therapy. Pembrolizumab is an immunotherapy that works by harnessing the body’s immune system to recognize and target cancer cells more effectively and is indicated for the treatment of adult and pediatric patients with unresectable or metastatic tumors exhibiting microsatellite instability-high (MSI-H) or a mismatch repair deficient (dMMR) biomarker. The approval was granted based on pooled data from single‐arm studies in which the observed overall response rate was 33% (95% CI, 23.7–44.1) and majority of responding patients (78%) had response durations of 6 months or longer. Following the success of pembrolizumab, other tumor-agnostic therapies such as larotrectinib (Vitrakvi) and entrectinib (Rozlytrek) for cancers with NTRK gene fusions, and dabrafenib (Tafinlar) in combination with trametinib (Mekinist) for cancers with BRAF V600 mutation have gained approval. These approvals underscore a shift towards therapies designed to target biomarkers present across multiple cancer types (regardless of their location or histology), thereby benefiting a wider patient population.
Comprehensive genomic profiling is a pivotal component in precision medicine
The growing number of approved targeted therapies and immunotherapies in recent years has necessitated development of methods capable of analyzing a broad range of biomarkers rapidly and efficiently. Comprehensive genomic profiling (CGP) using next-generation sequencing (NGS) has emerged as a pivotal component in precision medicine, enabling assessment of hundreds of both common and emerging biomarkers including tumor mutational burden (TMB) and MSI from a single sample with high specificity and sensitivity. The integration of biomarker-directed therapies as standard of care in cancer management has the potential to significantly improve patient outcomes and reduce side effects commonly associated with chemotherapies.
* Canary Oncoceutics has a steadfast commitment to three fundamental pillars: advancing scientific knowledge, fostering collaboration, and ultimately, enhancing the lives of cancer patients worldwide. From cutting-edge research to impactful clinical advancements, Canary Oncoceutics aims to illuminate the transformative potential of tailored cancer treatments. Join us on this journey towards a future where every cancer patient receives personalized, effective treatment tailored to their unique needs. *
To know more, connect with us on LinkedIn, Facebook, Twitter, Instagram, and our YouTube Channel