Patients with early-stage breast cancer can avoid certain surgeries
A study published on December 12th, 2024 in the New England Journal of Medicine suggests that early-stage breast cancer patients undergoing breast-conserving surgery may safely skip sentinel lymph node biopsy (SLNB), the standard of care for axillary node staging of patients with early-stage breast cancer. The 5-year invasive disease-free survival rate were found to be similar in both groups (with and without SLNB), at around 92%. Data from this study suggests that patients can be spared from SLNB procedure, if the absence of pathological information does not affect the postoperative treatment plan.
Another study published at the same time in the Journal of the American Medical Association, indicates that more frequent monitoring may be a better approach instead of immediate surgery in patients diagnosed with low-risk ductal carcinoma in situ (DCIS). Although not all DCIS progresses to invasive cancer, DCIS is conventionally treated with surgery, frequently combined with adjuvant therapy. This two year follow-up study found that the rates of invasive cancer were low with no significant differences between active monitoring group (4.2%) and surgery group (5.9%). While the researchers continue to follow these patients over the next decade, these preliminary findings indicate that low-risk DCIS patients can potentially opt for active monitoring without worrying about increased risk of invasive cancer.
Advantages of analyzing circulating tumor DNA in breast cancer management
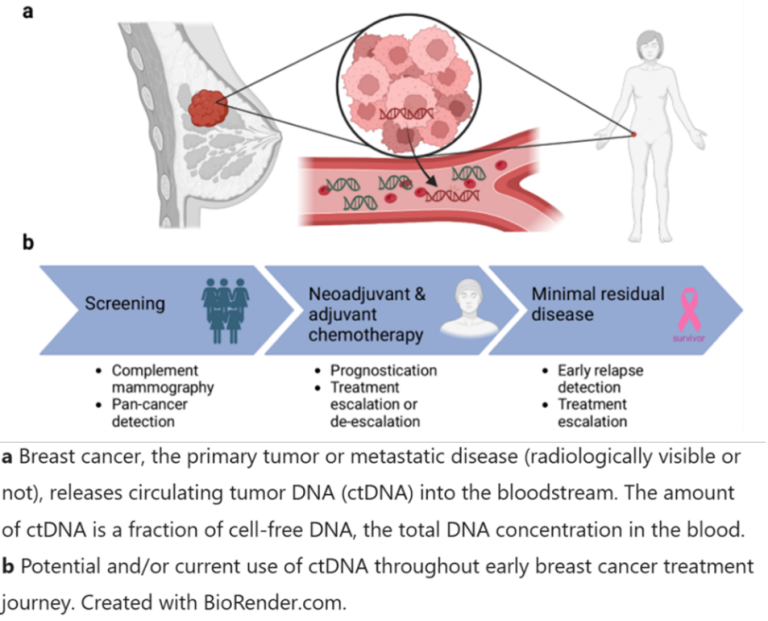
Traditionally, breast cancer diagnosis and treatment are based on American Joint Committee on Cancer’s tumor-node-metastasis (TNM) staging system, which is heavily reliant on imaging tests. Surgery is typically the usual treatment for early-stage breast cancer, which may be combined with adjuvant therapy. However, there is increasing concern that this standard approach has led to overdiagnosis and overtreatment, as highlighted by these two recent studies. Hence, there is a pressing need for better prognostic biomarkers to achieve a well-balanced risk-benefit ratio in breast cancer management. Circulating tumor DNA (ctDNA), which consists of small fragments of DNA that are shed from tumor cells into the bloodstream, has emerged as a promising biomarker. The use of plasma ctDNA is already gaining popularity in many advanced cancers including advanced breast cancer, for its many advantages: minimally-invasive, ease of serial samplings, rapid turnaround times, better detection of resistance mutations and ability to provide a real-time snapshot of tumor heterogeneity. But its use in early-stage breast cancer has been limited, most likely due to lower levels of ctDNA shedding in this setting.
Potential applications of ctDNA in early cancer detection and minimal residual disease monitoring
Recent advances in sequencing technologies and bioinformatics have sparked interest in using ctDNA for early-stage breast cancer detection. Although mammography is crucial for breast cancer detection and reduces breast cancer mortality by up to 20%, it can yield false-positive results in up to 12% of cases and false-negative results in up to 20% of cases. Multiple studies have indicated that ctDNA can be detected earlier, sometimes even months before tumors are detected through imaging or physical examinations, which enables earlier intervention when the disease is most treatable. Additionally, ctDNA shows promise in monitoring minimal residual disease (MRD) post-treatment. In early-stage breast cancer, the primary goal during postoperative follow-up is early detection of locoregional recurrence in the affected or contralateral breast, and typically does not involve detection of asymptomatic distant metastases. Traditional approaches (such as imaging tests and analysing tumor markers like carcinoembryonic antigen) have limited impact in the overall survival or quality of life. Since ctDNA can detect small tumor remnants in the bloodstream post-treatment, it can be used to detect early recurrences before relapse to guide treatment decisions in asymptomatic patients.
Although ctDNA holds great potential in early-stage breast cancer management, false-negative results remains a key challenge impeding its wide adoption in clinical practice. With ongoing efforts to improve sensitivity and reliability of ctDNA testing, technological advancements and more clinical trials, ctDNA testing could eventually become an integral component in breast cancer management.
Canary Oncoceutics has a steadfast commitment to three fundamental pillars: advancing scientific knowledge, fostering collaboration, and ultimately, enhancing the lives of cancer patients worldwide. From cutting-edge research to impactful clinical advancements, Canary Oncoceutics aims to illuminate the transformative potential of tailored cancer treatments. Join us on this journey towards a future where every cancer patient receives personalized, effective treatment tailored to their unique needs.