Tumor Mutational Burden (TMB)
TMB refers to the total number of mutations present within the coding region of a tumor genome. These somatic mutations (which are not inherited but acquired during tumor development) can be point mutations, insertions or deletions, copy number variations, and other genetic alterations. TMB is typically measured using next-generation sequencing (NGS), which detects multiple mutations across large coding region within a tumor sample. Although TMB is traditionally obtained via sequencing tissue samples, advancements in technology now also allows TMB to be calculated by sequencing circulating tumor DNA (ctDNA) from blood samples, known as blood TMB (bTMB).
TMB measurement
There are two primary methods to assess TMB: whole exome sequencing (WES) and targeted gene sequencing. WES involves sequencing the entire coding region of the tumor genome and provides a comprehensive view of mutations present in the tumor’s DNA. Despite WES being the current gold standard, many clinical labs use large, targeted gene panels that focus on a predefined set of cancer driver genes to measure TMB in routine diagnostics. Although this approach may be less comprehensive than WES, it is a more cost-effective and quicker method to assess TMB, while maintaining a high level of concordance with WES-based TMB results. A recent study published in 2024 showed that panel sizes beyond 1.04 Mb (megabase) and 389 genes are necessary to establish an accurate TMB calculation. TMB results are typically reported as mutations per megabase (mut/Mb) of DNA analyzed, and tumors with ≥10 mut/Mb are generally considered as TMB-High (TMB-H).
Importance of TMB
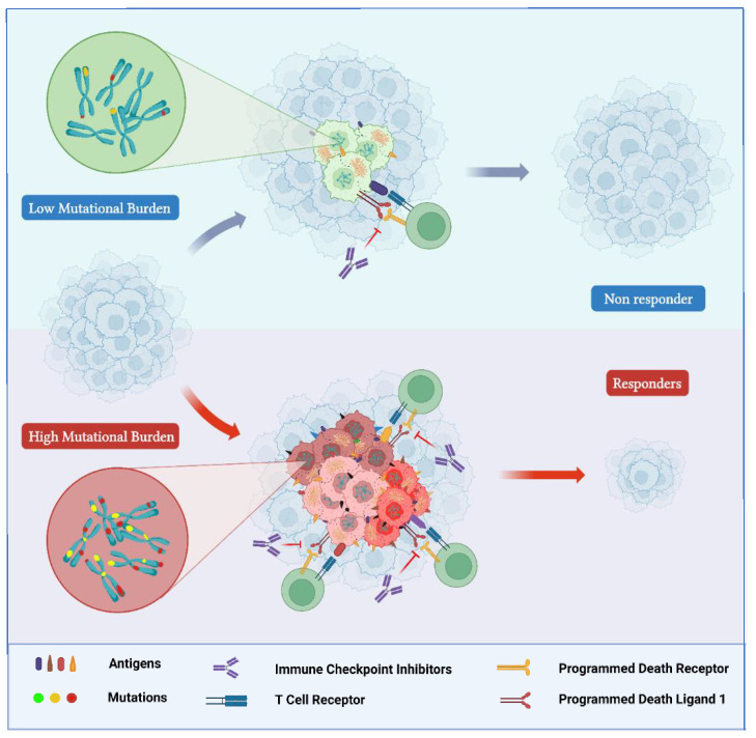
TMB is a U.S. FDA-approved biomarker to predict response towards immunotherapy in a variety of cancers such as non-small cell lung cancer, melanoma, bladder, and colorectal cancers. When a cell undergoes mutation, it may produce abnormal proteins known as neoantigens that are not found in normal tissues. These neoantigens are “foreign” to the immune system and can trigger an immune response. Immune checkpoint inhibitors (ICIs) such as pembrolizumab are a type of immunotherapy that block checkpoint proteins, thereby enhancing the immune system’s ability to recognize and attack cancer cells. Tumors with high TMB are more likely to be present more neoantigens, suggesting a higher likelihood of responding to ICIs. However, it is important to note that not all TMB-H tumors will respond to ICIs positively and vice-versa. Other factors such as PD-L1 expression, microsatellite instability (MSI) and tumor microenvironment, may also affect immunotherapy response. Hence, TMB should not be used as the sole definitive predictor in immunotherapy, and it is most useful when used in combination with other biomarkers and clinical information.
Canary Oncoceutics has a steadfast commitment to three fundamental pillars: advancing scientific knowledge, fostering collaboration, and ultimately, enhancing the lives of cancer patients worldwide. From cutting-edge research to impactful clinical advancements, Canary Oncoceutics aims to illuminate the transformative potential of tailored cancer treatments. Join us on this journey towards a future where every cancer patient receives personalized, effective treatment tailored to their unique needs.