Next-generation sequencing (NGS) has revolutionised cancer care and is becoming ubiquitous across the cancer care continuum. Multiple studies have demonstrated the real-world, positive impact of NGS testing on improving patient outcomes through shortening time to results, facilitating tailored treatment strategies and better patient selection for clinical trials. The rapidly growing field has led to the commercialisation of NGS tests using a variety of gene panels that differ in size, gene content, mutation types that can be detected, and others. Hence, it is important to evaluate the factors that can impact the comprehensiveness, accuracy, quality and relevance of the test when comparing between different NGS tests.
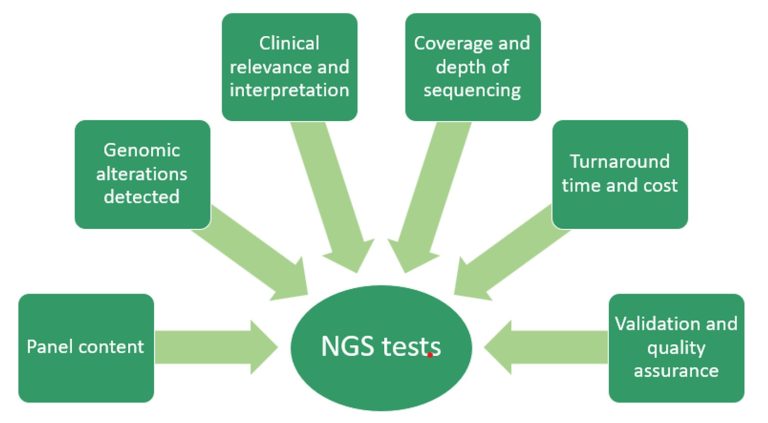
- Panel content
- Genes included: Check which genes are included in the panel. A comprehensive panel should consist of genes associated with key oncogenic pathways, including oncogenes, tumor suppressor genes and DNA repair genes. The gene list should be relevant to the tumor type analyzed (e.g., breast, colorectal, lung cancer) and aligned with clinical guidelines (e.g., National Cancer Comprehensive Network [NCCN], European Society for Medical Oncology [ESMO]).
- Panel size: Size of the panel has important impact for certain biomarkers like tumor mutational burden (TMB). Smaller panels can result in imprecise measurement of TMB, whereas larger panels show significantly smaller variance, giving greater accuracy in results. Since larger panels can incur higher cost, there is a need to optimize the cost-benefit ratio. A recent study published in 2024 showed that panel sizes beyond 1.04 Mb (megabase) and 389 genes are necessary for the basic discrete accuracy in calculating TMB.
- Genomic alterations detected
- Multiple classes of genomic alterations: The NGS test should be able to detect a broad range of genomic alterations, including single-nucleotide variants (SNVs), small insertions and deletions (InDels), copy number variations (CNVs) and structural variants (SVs) to provide a comprehensive view of the tumor profile.
- Genomic signatures: Ideally, the NGS test should also include assessment for genomic signatures as TMB and microsatellite instability (MSI), which are pivotal in predicting response towards immunotherapies.
- Clinical relevance and interpretation
- Actionable mutations: The NGS test should not only be able to detect mutations, but should also provide clear clinical insights regarding the actionability of these mutations by including recommendations on therapies based on clinical guidelines (e.g., NCCN, ESMO). In addition, it would also be informative if the test report includes clinical trial opportunities based on a tumor’s profile.
- Interpretation support: Some companies provide value-added services, which includes support to help oncologists interpret the complex molecular data contained within the report and select best treatment options for their patients.
- Coverage and depth of sequencing
- Coverage: Assess whether the panel design provides adequate coverage of gene targets and critical exons are sufficiently covered to ensure complete analysis of relevant mutations.
- Sequencing depth: Depth of sequencing refers to the number of times a specific base (nucleotide) is read during sequencing. Higher sequencing depth (e.g., 1,000× or more) improves test sensitivity and ability to detect rare variants at low allele frequencies (e.g., 1% variant allele frequency [VAF]).
- Turnaround time and cost
- Turnaround time: Different NGS testing laboratories will have varying processing times, which can be affected by a test’s complexity and the laboratory workflow. Nevertheless, quicker test results (e.g., within 7-14 working days) are critical in clinical settings to guide timely decision-making.
- Cost: Consider the cost of the NGS test relative to the range of genes tested and clinical information provided. Some NGS tests using less comprehensive gene panels may be cheaper, but may miss important actionable mutations, leading to sup-optimal treatment options.
- Validation and quality assurance
- Clinical validation: The NGS test should already be clinically validated with analytical and clinical performance metrics (high accuracy, sensitivity and specificity).
- Quality assurance: To ensure that the NGS test results are of high quality and reliability, look for NGS tests that are Clinical Laboratory Improvement Amendments (CLIA) certified and College of American Pathologists (CAP) accredited.
* Canary Oncoceutics has a steadfast commitment to three fundamental pillars: advancing scientific knowledge, fostering collaboration, and ultimately, enhancing the lives of cancer patients worldwide. From cutting-edge research to impactful clinical advancements, Canary Oncoceutics aims to illuminate the transformative potential of tailored cancer treatments. Join us on this journey towards a future where every cancer patient receives personalized, effective treatment tailored to their unique needs. *
To know more, connect with us on LinkedIn, Facebook, Twitter, Instagram, and our YouTube Channel