DNA mismatch repair
Microsatellites are short, tandem repetitive sequences that are highly susceptible to transcription errors (base mismatches and small insertions or deletions) during DNA replication. Key proteins involved in DNA mismatch repair (MMR) such as MLH1, MSH2, MSH3, MSH6, MLH2, MLH3, PMS1, PMS2, and EPCAM are responsible for detecting and repairing these errors. Cells with functional repair proteins are considered to be microsatellite stable (MSS) and are able to repair errors proficiently. On the other hand, microsatellite instability (MSI) occurs as a result of deficiency within MMR proteins. Cells with high level of MSI (also called MSI-High, MSI-H or deficient mismatch repair, dMMR) are unable to repair errors proficiently, leading to accumulation of replication errors and alteration in the length of microsatellites.
MSI and MMR testing
MSI detection is typically performed on tumor tissue using several methods: immunohistochemistry (IHC), polymerase chain reaction (PCR), and next-generation sequencing (NGS). IHC detects the presence or absence of MMR proteins quickly and easily. An absence or reduced MMR protein expression in tumor tissue compared to normal tissue indicates a loss of MMR function, which is often associated with MSI. However, MMR protein expression does not necessarily provide a conclusive measure of MSI because in some cases with missense mutations in MMR genes, the corresponding MMR protein expression remains intact but is functionally inactivated (5-10% of non-functional proteins retain antigenicity). PCR is the current accepted gold standard to determine MSI status, whereby microsatellites in tumor DNA are amplified and compared to the normal tissue DNA. A difference in the size of the microsatellites indicates MSI. Although PCR can analyze multiple markers simultaneously and directly measure changes in DNA caused by loss of MMR proteins, it is still limited by the number of microsatellite markers used and may not detect all forms of MSI. Hence, NGS has emerged as a more comprehensive approach to detect MSI, as it can evaluate a large number of microsatellite loci across the genome and also detect lower levels of MSI that might be missed by other methods. Significant differences (as determined by NGS algorithms) in the microsatellite sequence length between tumor tissue and matched normal tissue (or a consensus sequence) indicates MSI. Besides detecting MSI, NGS also provides additional information such as mutations, copy number variations and other genomic alterations within the same assay. MSI can also be determined in circulating tumor (ctDNA), which is fragmented DNA released by cancer cells into the blood, in the absence of tissue specimens.
MSI as a biomarker
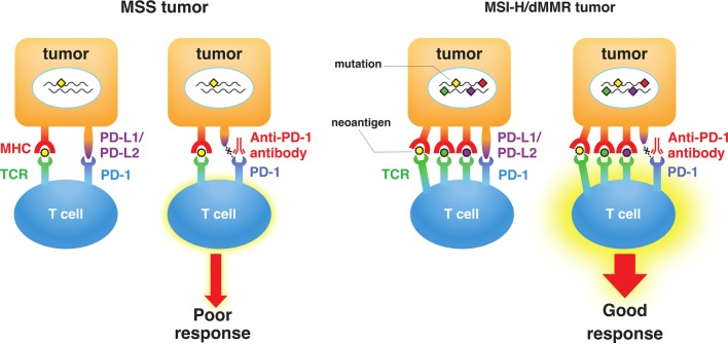
Historically, MSI has been used as a biomarker since 1993 for hereditary nonpolyposis colorectal cancer (HNPCC), also known as Lynch syndrome. Lynch syndrome is an autosomal dominant disorder caused by hereditary mutations (germline mutations) in the MMR genes, resulting in dMMR. Although it is more prevalent in colorectal cancer, Lynch syndrome is also associated with increased risks in other cancers including endometrial, ovarian and gastric cancer. In a paradigm shift for cancer treatment, MSI was approved as the first tumor-agnostic biomarker in predicting response towards immunotherapy in 2017. Tumors with MSI-H (as determined by PCR or NGS) or dMMR (as determined by IHC) respond better to immunotherapies, such as immune checkpoint inhibitors (ICIs). Tumors with defective MMR proteins have an increased number of somatic mutations that produce neoantigens. As a result, these tumors are more effective at priming an immune response, leading to better immunotherapy response.
Canary Oncoceutics has a steadfast commitment to three fundamental pillars: advancing scientific knowledge, fostering collaboration, and ultimately, enhancing the lives of cancer patients worldwide. From cutting-edge research to impactful clinical advancements, Canary Oncoceutics aims to illuminate the transformative potential of tailored cancer treatments. Join us on this journey towards a future where every cancer patient receives personalized, effective treatment tailored to their unique needs.